|
Then they asked me to return home, get a gallon of water and bring it to them to quench their thirst. It was a hot summer day, and they went up this nearby hill to bury their collection of bottle caps. There was a time in my life when I could use this concept of water density to get back at my older brother and his friends for always picking on me. One example that can be made for this is molten silica. 
This phenomenon is usually referred to as ‘negative thermal expansion’ and applies to those bonded with intermolecular forces and other chemical composition factors. There are cases where it is usually the other way around, where further cooling of a substance decreases its density.But the limit ends at 4☌, where pure water is at its most dense. Of course, this is true for other substances as well. When water is cooled at room temperature, it becomes denser over time.Temperature can also change water density, but the correlation is still unclear and inconsistent.The density of water is approximately 1 gram per cubic centimeter (1 g/cm3) or 1 gram per milliliter (1g/mL).The following are the common factors that affect the density of water. It is simply just its weight for every specific volume.įor example, water’s density under normal conditions is 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit), to be specific but is rounded off to 1 gram per mL or cubic centimeter (1g/mL or cc) for uniformity. In other words, if an object occupies space and has mass, its density can also be measured. Mass and weight are often misused because of the confusion about their respective definitions, but since we are only referring to one gravity of Earth, mass and weight are pretty much the same.ĭensity is a constant factor in all matter that is composed of molecules. That is the technical definition of the term. Propane (sea level, 20 ☌) - 1.The sentence “Density is the mass per unit volume of a substance” is always mentioned in class.Carbon monoxide (sea level, 20 ☌) - 1.165 kg/m 3.Carbon monoxide (sea level, 0 ☌) - 1.250 kg/m 3.Carbon dioxide (sea level, 20 ☌) - 1.842 kg/m 3.Carbon dioxide (sea level, 0 ☌) - 1.977 kg/m 3.Walnuts, hazelnuts, grounded - 520 kg/m 3.Here are the products and their densities available in our calculator: That's it! The volume to mass calculator will find the result in less than a second! Change the unit first before you enter a number. Second, input volume or mass in the correct unit. Check if the material is on our list or input known density. In our volume to mass calculator, you can input values in any unit you want - our calculator will convert them and make correct calculations.įirst, you need density. 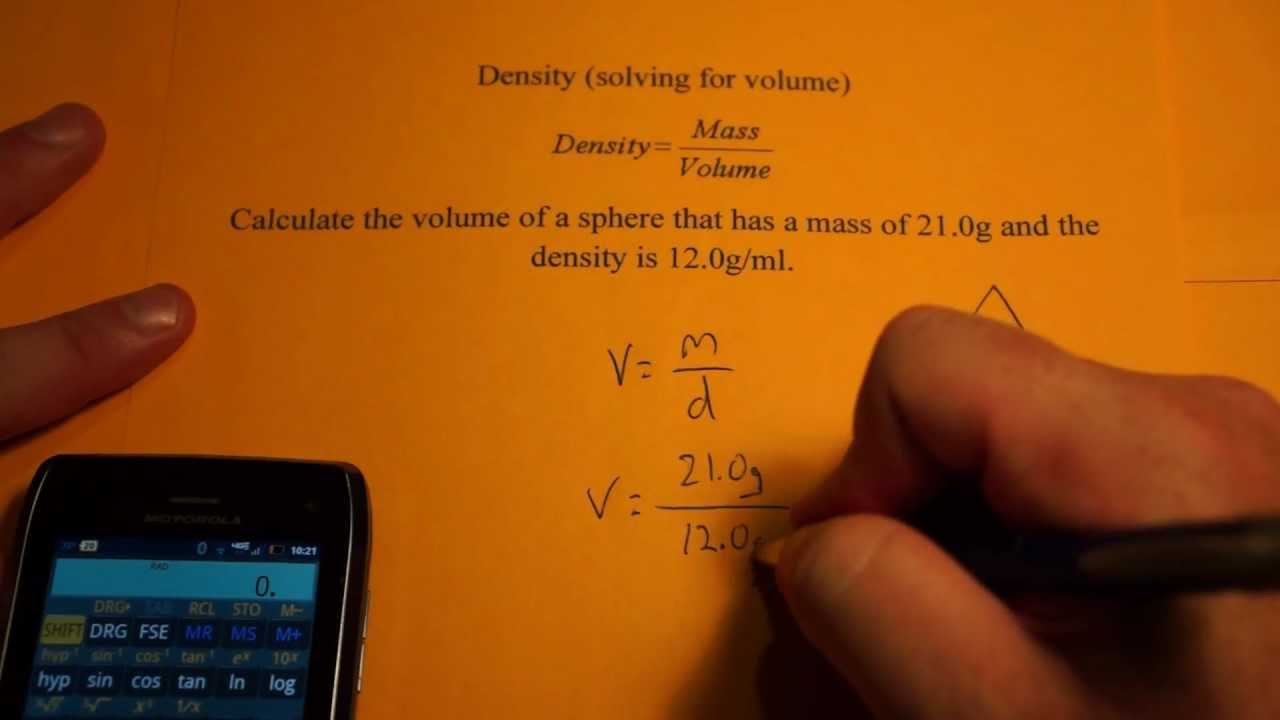
For example, if you input mass in pounds and volume in gallons, you will get density in pounds per gallon. Whenever you use this formula, remember to be concise with units. So, how do you calculate volume with mass and density? You need to rearrange the formula to: While it doesn't matter in most cases (like cooking), it might be significant if you're building something or doing a science experiment. It's worth knowing that density slightly changes with temperature and pressure. You've probably already guessed what formula will tie these three values together: It means that one liter of water weighs one kilogram. For example, water at 4 ☌ has a density of 1 kg/l. 
What is density? It's mass-to-volume ratio - a physical property of every material. 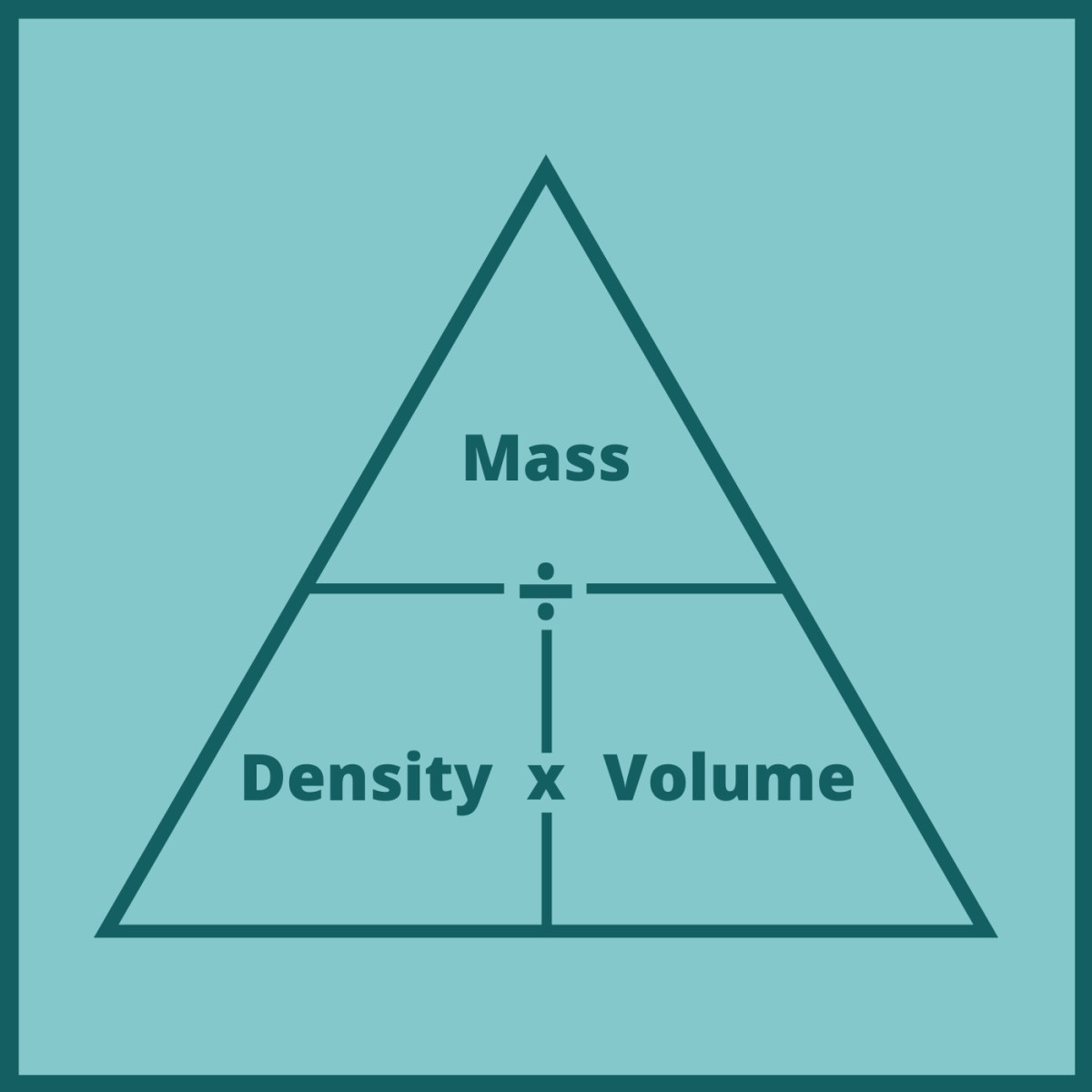
To convert something's mass to volume or vice versa, you need to know its density.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
